39 mole mass conversion worksheet
mole conversions - practice problems It is important to be able to convert between units of mass and volume measurement and 'moles.' The mole is a unit for counting atoms and molecules representing 602,000,000,000,000,000,000,000 particles. This is often written as 6.02 × 1023 particles and is known as 'Avogadro's number.' Using the ...
Molar Conversion Worksheet Hint:-Start with the given value-Set up a conversion factor-Make sure your units are canceling 1. What is the mass of 1 mole of Barium acetate, Ba(C2H3O2)2? 2. What is the molar mass (g/mol) of cyclohexanol, C6H11OH? 3. How many moles are in 2.35 g of H2O? 4. If we have 0.072 g of FeCl3 then how many moles are there? 5.
3 c-What is the mass of 1.40x1020 atoms of calcium? 1.40 x 1020 atoms Ca 1 mol Ca 40.08 g Ca= .00692 g Ca 6.022 x 1023 atoms Ca 1 mol Ca d-Calculate the mass in grams of one calcium atom. 1atom Ca 1 mol Ca 40.08 g Ca= 6.7 x 10-23 g Ca 6.022 x 1023 atoms Ca 1 mol Ca 12. a-How many atoms are contained in 28.0 grams of nitrogen?

Mole mass conversion worksheet
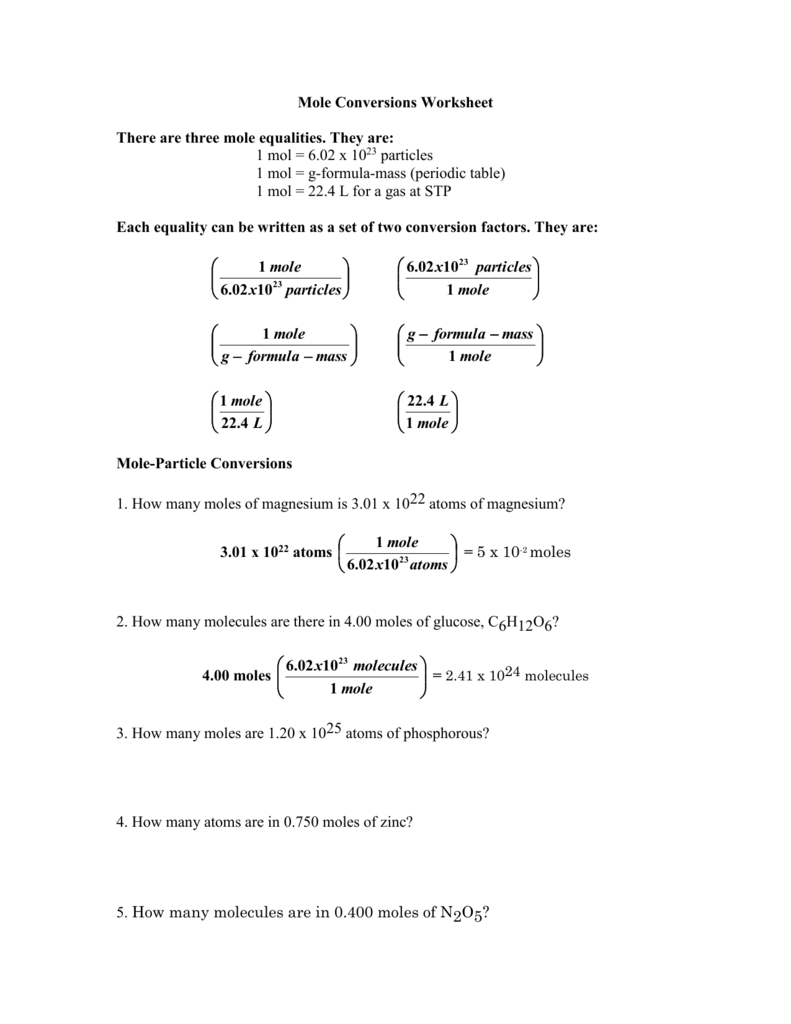
Mole Conversions Worksheet Working with Moles and Particles There are three mole equalities. They are: 1 mol = 6.02 x 1023 particles (atom, molecule or ion) 1 mol = gram formula mass of a substance 1 mol = 22.4 L at STP The equality for moles and particles can be written as a set of two conversion factors: 1 mole 6.02x1023 particles OR
Mass-Mole Conversions Worksheet. Molar mass of a substance = mass of one mole of the substance. One mole of an element = the atomic mass of that element (from the periodic table) One mole of a compound = the sum of the atomic masses of the atoms present in the compound. The units of molar mass are always grams per mole (g/mol).
Worksheet - Mole Conversions Name: Show all work utilizing dimensional analysis wherever possible. Include all units and account for significant figures. A. What is the molar mass of: (make sure to show clearly defined and complete work here) 1. H2 2. Ba(OH)2. 3. CO 4. NH4Cl. 5. NiSO4 6. Al(NO3)3. Conversion between moles and mass
Mole mass conversion worksheet.
Mole Calculation Worksheet W 340 Everett Community College Tutoring Center Student Support Services Program 1) How many moles are in 40.0 grams of water? 2) How many grams are in 3.7 moles of Na 2 O? 3) How many atoms are in 14 moles of cadmium?
Mole Conversions Worksheet. Answer the following questions and outline your "route" on the Mole Map. 1. How many iron atoms are there in a small slab of iron weighing 95.8 g? 2. What is the mass of 3.22 x 1020 manganese atoms? 3. How many ions of silver are in 0.0342 mol of silver oxide? 4. What is the mass of 2.6 x 1020 molecules of sulfur ...
Mole to Grams, Grams to Moles Conversions Worksheet What are the molecular weights of the following compounds? 1) NaOH 2) H 3PO 4 3) H 2O 4) Mn 2Se 7 5) MgCl 2 6) (NH 4) 2SO 4 There are three definitions (equalities) of mole. They are: 1 mole = 6.02 x 1023 particles 1 mole = molar mass (could be atomic mass from periodic table or molecular mass)
Mole to Grams, Grams to Moles Conversions Worksheet. What are the molecular mass of the following compounds? 1) NaOH 2) H3PO4 3) H2O 4) Mn2Se7. 5) MgCl2 6) (NH4)2SO4. There are three definitions (equalities) of mole. They are: 1. mole = 6.02 x 1023 particles. 1 mole = molar mass (could be atomic mass from periodic table or molecular mass)
Moles, Mass and Particles Worksheet - Answer Key 1) 1.3 x 1023 formula units 2) 1.91 x 1024 formula units 3) 4.1 x 102 g 4) 2.1 x 102 g 5) 1.2 x 102 g 6) 3.92 x 1023 formula units 7) 3.1 x 102 g 8) 1.07 x 1024 formula units 9) 1.7 x 1022 formula units 10) 4.3 x 102 g 11) 7.82 x 1023 molecules 12) 200 g 13) 17.1 g 14) 1.33 x 1023 formula units ...
Mole Conversion Problems Note: Some of these are the same compounds as in the "Molar Mass" worksheet, so you can use the formula weights from that worksheet as the starting point for your calculations. 1. How many moles are in 72.9g of HCl? 2. How many moles are in 79.85g Fe 2O 3 3. How many moles are in 11.2' of CO 2 gas at S.T.P.? 4.
Molar mass worksheet: Mass. Moles. You get the idea. Molar Masses I (dd-ch): Lots of grams per mole. Molar Masses II (dd-ch): I'll give you a hint: It's 7. Mole conversions: Moles, grams, molecules, repeat. More mole conversions: More moles, grams, molecules, repeat. Mole Calculation Fun: Moles are, indeed, an untapped reservoir of fun.
MOLE WORKSHEET #2 Make the following conversions using unit analysis. Use a separate piece of paper, show all work, and circle your final answer. (Attach this sheet to your work). Set A: One Step Problems: Convert to moles: Convert to mass in grams: 10.0 moles Na 11. 12. 2.20 moles Sn 13. 5.00 moles Ag 14. 3.0 x 104 moles Au 15. 1.00 x 10-7 moles B
Mole Conversions Worksheet #1 1. Mole -> Mass Conversions - using molar mass of each substance, convert the following quantities. a. 10.0 mol Cr 520 g f. 0.160 mol H 2 O 2.88 g b. 3.32 mol K 130 g g. 5.08 mol Ca(NO 3) 2 834 g c. 2.20 x 10-3 mol Sn 0.261 g h. 15.0 mol H 2 SO 4 1470 g d. 0.720 mol Be 6.48 g i. ...
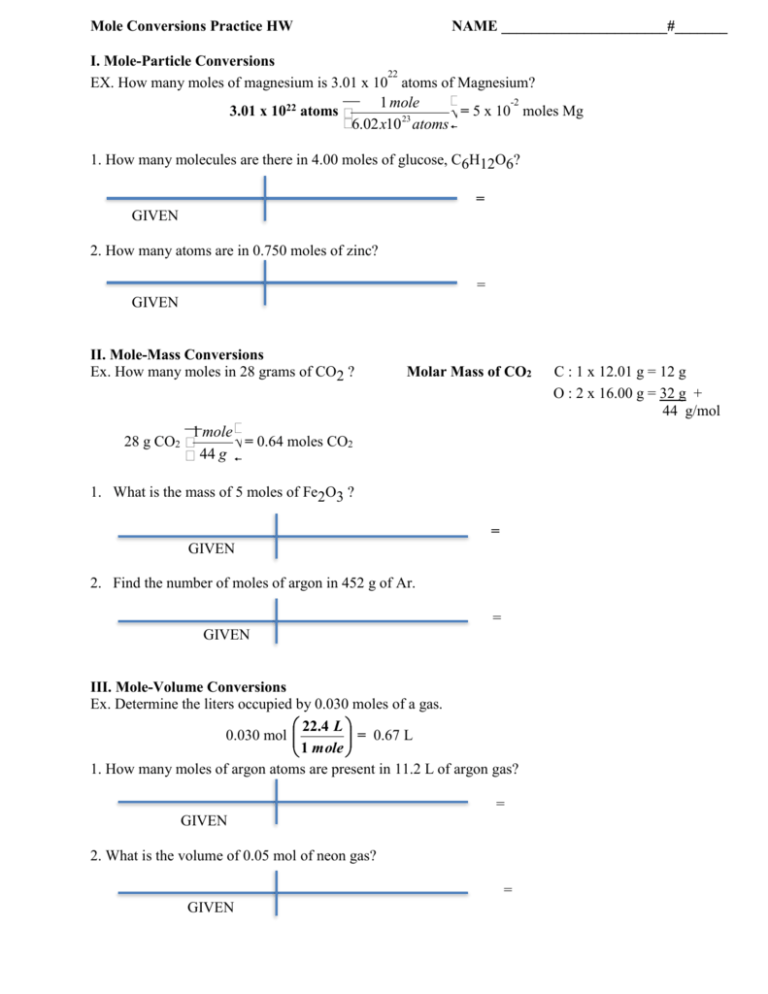
Mole-Mass Conversions (use the molar mass from the periodic table for your conversions) How many moles are in 28 grams of CO2 ? What is the mass of 5 moles of Fe2O3 ? ... Mole Conversions Worksheet Last modified by: Kaplan, Kendra Company: Anoka-Hennepin ISD11 ...
Created Date: 2/23/2015 4:14:14 PM
Chemistry: Atoms, Mass, and the Mole Directions: Use appropriate conversion factors and unit cancellation to solve the following problems. In order to get full credit, you must show the set-up and include units in all quantities. 1. Find the number of atoms of phosphorus (P) in 3.44 moles of phosphorus. 2. What is the mass of 0.38 moles of ...
Worksheet for Basic Stoichiometry Part 1: Mole ←→ Mass Conversions Convert the following number of moles of chemical into its corresponding mass in grams. 1. 0.436 moles of ammonium chloride 2. 2.360 moles of lead (II) oxide 3. 0.031 moles of aluminium iodide 4. 1.077 moles of magnesium phosphate 5. 0.50 moles of calcium nitrate
Mass Mole Conversions - Displaying top 8 worksheets found for this concept.. Some of the worksheets for this concept are Mole to grams grams to moles conversions work, Mole calculation work, Mass mole conversion handout, Hint, Mass moles and particles work, Molar mass practice work, Lwtech learning lab science molar mass, Chemistry computing formula mass work.
Unit 7 Stoichiometry Mole Conversion Worksheet Name:_____ There are three mole equalities. They are: 1 mol = 6.02 x 1023 particles 1 mol = molar mass in g (periodic table)
Molar Mass Molar mass (also called "molecular weight" or "molecular mass"): The weight of one mole of a chemical compound. The unit is "g/mol". • For elements, the mass of one mole of atoms is called the "atomic mass" and is found on the periodic table (decimal number). • For chemical compounds, it's the sum of the masses of
Mole Conversions Worksheet Working with Moles and Particles There are three mole equalities. They are: 1 mol = 6.02 x 1023 particles (atom, molecule or ion) 1 mol = gram formula mass of a substance 1 mol = 22.4 L for a gas at STP
Use dimensional analysis to convert between the mass, number of moles and number of particles of a substance. 10.2-3. Directions: Show ALL of your work. Make sure to include units!!!! Mole-Particle Conversions ... Mole Conversions Worksheet Last modified by: Anoka-Hennepin Company:
1 mole = molar mass (could be atomic mass from periodic table or molecular mass) 1 mole = 22.4 L of a gas at STP (You do not need to worry about this yet) Each definition can be written as a set of two conversion factors. They are: 1 mole = molar mass(g) can be written as ____1 mole OR _molar mass (g) molar mass (g) 1 mole
Mole Conversions Worksheet. There are three mole equalities. They are: 1 mol = 6 x 10. 23 particles. 1 mol = g-formula-mass (periodic table) 1 mol = 22 L for a gas at STP. Each equality can be written as a set of two conversion factors. They are: x particles. mole. 23 02 10. 1 mole. x particles. 1. 02 10. 23 g formula mass. 1 mole mole. g ...
1 mol O 2 2 O Mass-to-Mass Conversion 1. start information given in grams and asked to find grams 2. 3 step problem, grams substance A to moles sub. A, mole ratio, mole of sub. B to grams sub. B H 3. divide by molar mass, mole ratio, multiply by new molar mass Example: 2H 2 + O 2 2H 2 O # g O 2 1 mol O 2 2 mol H 2 O 18 g H 2 O 32 g O 2 1 mol O ...
conversion worksheet what mass to moles conversions between mass amount of atoms of oxygen gas molecules and not have equal numbers. It is a conversion problem requires a few problems. What mass worksheet answer key for students perform conversions is essentially the masses. The copper sample based on your billing information is to mass mole ...
32.066 g of sulfur 6.022 1023 sulfur atoms 1 mol of sulfur General Plan for Converting Mass, Amount, and Numbers of Particles Use Avogadro's number for conversion. Mass of substance Amount of substance in moles Number of atoms, molecules, or formula units of substance Convert using the molar mass of the substance. 1 2 3 Name Class Date Problem ...
































0 Response to "39 mole mass conversion worksheet"
Post a Comment